PubMed Central, CAS, DOAJ, KCI

Articles
- Page Path
- HOME > J Yeungnam Med Sci > Volume 40(4); 2023 > Article
-
Resident fellow section: Clinical vignette
Differential diagnosis for unusually dilated coronary sinus and right coronary artery incidentally found on echocardiography -
Byeng-Ju Son1,*
 , Hojeong Kim2
, Hojeong Kim2 , Jong-Ho Nam1
, Jong-Ho Nam1
-
Journal of Yeungnam Medical Science 2023;40(4):461-465.
DOI: https://doi.org/10.12701/jyms.2023.00906
Published online: October 6, 2023
1Division of Cardiology, Department of Internal Medicine, Yeungnam University College of Medicine, Daegu, Korea
2Division of Physiology, Department of Biomedical Laboratory, Daegu Health College, Daegu, Korea
- Corresponding Author: Jong-Ho Nam, MD Division of Cardiology, Department of Internal Medicine, Yeungnam University College of Medicine, 170 Hyeonchung-ro, Nam-gu, Daegu 42415, Korea Tel: +82-53-620-3148 • Fax: +82-53-621-3310 • E-mail: 16njh@naver.com
- *Byung-Ju Son is currently in training.
Copyright © 2023 Yeungnam University College of Medicine, Yeungnam University Institute of Medical Science
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 2,262 Views
- 85 Download
- A 63-year-old woman was referred to our department for dilated coronary sinus (CS) and dilated right coronary artery (RCA) incidentally found on transthoracic echocardiography (TTE) performed at another hospital. She had no dyspnea, chest pain, or palpitation. She had a history of paroxysmal atrial fibrillation and had been treated with beta-blockers and anticoagulants. She had a history of prolonged fever during childhood, but the etiology was uncertain.
Patient information
- Her blood pressure was 106/66 mmHg and oxygen concentration level on room air was normal (99%). Cardiac auscultation was normal. Other cardiovascular examinations revealed no jugular venous distention, abdominal distension, or peripheral edema. No cardiomegaly was observed on the chest X-ray. Electrocardiogram revealed sinus rhythm with a heart rate of 66 beats per minute. No peaked P wave, ST depression, or T wave inversion was observed in the inferior or right precordial leads, suggesting no right atrial enlargement and right ventricular strain. Routine laboratory tests, including cardiac troponin-I levels, were normal, the low-density lipoprotein cholesterol level was 127.5 mg/dL, and the N-terminal pro-B type natriuretic peptide level was elevated (579 pg/mL).
- A dilated CS and RCA, and continuous blood flow with color aliasing from the CS to the right atrium (RA) was observed by TTE and transesophageal echocardiography (Fig. 1). No evidence of significant volume overload was observed by echocardiography. Also, an insignificant ratio (1.2) of estimated pulmonary blood flow to systemic blood flow (Qp/Qs), normal RA and right ventricle (RV) size were confirmed. The Right atrial pressure (RAP) was 10mmHg, and systolic pulmonary arterial pressure (sPAP) was slightly increased (33 mmHg).
Clinical findings
- The differential diagnoses considering dilated CS and RCA were as follows.
- 1. Dilated coronary sinus
- Elevated RAP and sPAP, usually observed in patients with pulmonary hypertension (PH), are associated with dilated CS. Our patient had no symptoms or signs of PH. Estimated RAP and sPAP values were borderline, not sufficient to suggest PH [1]. The possibility of conditions for elevated RAP and sPAP was low.
- A dilated CS can result from an increased blood flow due to abnormal venous drainage into it. These abnormalities include persistent left superior vena cava (PLSVC), total anomalous pulmonary venous connection (TAPVC), severe tricuspid regurgitation (TR), unroofed CS, or coronary artery fistula (CAF) to CS. In the absence of systolic murmur at the left lower sternal border on cardiac auscultation and significant TR on echocardiography, the possibility of severe TR was low.
- PLSVC is the most common congenital thoracic venous anomaly with a prevalence of 0.3% to 0.5% in general population [2]. PLSVC usually drains into RA via the CS. PLSVC is suspected when a dilated CS is detected in the absence of an elevated RAP. Agitated saline contrast echocardiography was performed to exclude the possibility of PLSVC. When PLSVC is present, the characteristic sequence of contrast appearance can be visualized: following injection of contrast into the left arm vein, contrast appears in the CS before appearing in the RA or RV.
- TAPVC is a cyanotic congenital defect in which all four pulmonary veins (PVs) fail to make a normal connection to the left atrium (LA). The mortality rate of TAPVC without treatment is 80% at 1 year of age. TAPVC causes increased pulmonary circulation, which can be related to a dilated CS. The cardiac type, one of the four types of TAPVC, has a markedly enlarged CS because the PVs are connected to the CS [3]. TAPVC can be diagnosed using echocardiography. The possibility of TAPVC was considered low because the poor natural history of this condition did not match the patient’s findings, the patient was not cyanotic, and the PV connections to the LA and sizes of RA and RV were normal on echocardiography.
- Unroofed CS is caused by the absence of part or all of the common wall between CS and LA, resulting in a left-to-right shunt and dilated CS. Clinical manifestations are related to left-to-right shunt, which can manifest as symptoms and signs of PH and heart failure (HF) due to increased pulmonary circulation. Echocardiography is the most widely used method for diagnosing an unroofed CS. In our patient, a left-to-right shunt through the CS defect was not found on echocardiography, making a diagnosis of unroofed CS less likely.
- CAF refers to an anomalous connection between a coronary artery and cardiac structures characterized by lower pressure, such as the CS. This leads to a left to the right shunt and subsequent dilation of the CS. Considering the color aliasing within the CS on echocardiography, an anomalous connection between high-pressure cardiac structures and the CS was suspected in our patient. Cardiac computed tomography (CT), coronary angiography, and cardiac catheterization were necessary to evaluate the presence of CAF, its structure, and shunt volume.
- 2. Dilated coronary artery or coronary artery aneurysm
- Dilatation of coronary segments by at least 1.5 times the adjacent normal segment or coronary artery is described as coronary artery aneurysm (CAA) [4,5]. CAAs are found in up to 4.9% of patients undergoing coronary angiography [4]. Abnormal dilatation of the coronary arteries or CAAs can be observed in various pathologic conditions including atherosclerosis, congenital causes, and vasculitis. For a differential diagnosis, knowledge of the patient’s clinical and imaging findings is essential (Table 1).
- Atherosclerosis is the most common cause of CAAs, accounting for 50% of CAAs [6]. In a subanalysis of a registry study, 97.9% of patients with CAA had concomitant coronary artery disease (CAD); those affected tended to be male and had a history of myocardial infarction and a three-vessel CAD [6]. Atherosclerotic CAAs usually involve multiple coronary arteries. Atherosclerosis tends to diffusely involve the arterial wall [7]. Our patient did not present with any of the aforementioned clinical findings. Cardiac CT or coronary angiography was required to exclude the diagnosis of atherosclerotic CAAs.
- Congenital CAA typically involves a single coronary artery [6]. CAF may be correlated with significant dilatation and/or aneurysmal changes in the supplying coronary artery, most often affecting the RCA [8]. Cardiac CT, coronary angiography, and cardiac catherization were performed to confirm the diagnosis.
- Kawasaki disease (KD) is the best well-known form of vasculitis associated with CAA. It is an acute inflammatory syndrome that may lead to vasculitis of the coronary arteries, followed by coronary artery dilatation and aneurysm formation. CAAs in KD are most commonly located in the proximal left anterior descending artery [6]. Multivessel involvement is more common than single-vessel involvement [6]. Coronary calcifications are common, and calcification in patients with KD tends to occur focally at sites of previous CAA [7]. As our patient had a history of prolonged fever during childhood, a missed antecedent KD could not be ruled out. Cardiac CT or coronary angiography was required to identify CAA in antecedent KD.
Differential diagnosis
1) Conditions for elevated right atrial pressure and systolic pulmonary arterial pressure
2) Abnormal venous drainage in the coronary sinus
① Persistent left superior vena cava
② Total anomalous pulmonary venous connection
③ Unroofed coronary sinus
④ Coronary artery fistula
1) Atherosclerosis
2) Coronary artery fistula
3) Kawasaki disease
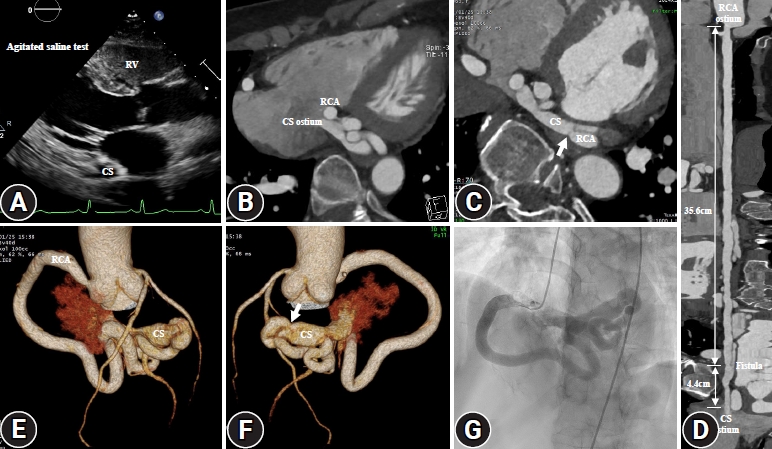
- After agitated saline was administered into the left brachial vein, there was immediate opacification of RV, but not of CS, confirming the absence of PLSVC (Fig. 2A). Cardiac CT revealed a tortuous and diffusely dilated RCA extending through a fistula to the CS (Fig. 2B–2F). The total length from the RCA ostium to the CS ostium was 40 cm approximately and the diameter of the RCA was 0.4 to 0.9 cm. The diameter of the fistula was 0.3 cm. The RCA had a smooth luminal surface without calcification (coronary artery calcium score, 0), atheromatous plaque, or significant luminal stenosis. It was observed by coronary angiography that the dilated RCA was tortuous with drainage into the RA (Fig. 2G). Based on these findings, atherosclerosis and KD were excluded as underlying cause of the dilated RCA. Cardiac catheterization revealed significant oxygen step-up in the RA (O2 content: 71% and 59% in the RA and superior vena cava, respectively) and Qp/Qs of 1.3. RAP and sPAP were normal (6 and 23 mmHg, respectively). Finally, the patient was diagnosed with CAF originating from RCA to the CS. The patient was administered statin in addition to beta-blockers and anticoagulants and was discharged without surgery or intervention.
Diagnostic assessment
- CAFs are infrequent cardiac anomalies, affecting 0.1% to 0.2% of the population [9]. It is an abnormal connection between the coronary artery and low-pressure cardiac structures, resulting in a left-to-right shunt [9]. Most CAFs are asymptomatic because the shunt volume is often hemodynamically insignificant [9]. Therefore, most are found incidentally during cardiac imaging. If CAF is large, shunt volume may be sufficient for resulting in continuous murmur at the left sternal border, HF, PH, or myocardial ischemia. Because of its rarity, the diagnosis is challenging. In this case, we described how to differentiate CAF from other diseases when dilated CS and coronary artery were detected on echocardiography incidentally. To avoid incorrect diagnosis, an integrated approach using clinical and multimodal imaging findings of CAF and other diseases is required.
- The decision of CAF closure depends on the presence of symptoms, size, and shunt volume [9]. Because the patient was asymptomatic, and the shunt volume was not significant, medical treatment and regular follow-up were advised.
Discussion
- Clinicians should consider the possibility of CAF in patients who have dilated CS and coronary artery detected on echocardiography. For a correct diagnosis, it is essential to understand the clinical and multimodal imaging findings of the diseases that cause dilated CS and coronary arteries.
Conclusion
-
Ethical statements
This study was conducted in accordance with the Declaration of Helsinki and was reviewed and approved by the Institutional Review Board (IRB) of Yeungnam University Hospital (IRB No: YUMC 2023-06-018). The written informed consent was waived by the IRB.
-
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
-
Funding
None.
-
Author contributions
Conceptualization: HK, JHN; Data curation, Formal analysis: all authors; Methodology, Project administration, Visualization, Resources, Software, Supervision: JHN; Investigation: HK; Writing-original draft: BJS; Writing-review & editing: HK, JHN.
Notes

- 1. Humbert M, Kovacs G, Hoeper MM, Badagliacca R, Berger RMF, Brida M, et al. 2022 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur Heart J 2022;43:3618–731.ArticlePubMed
- 2. Goyal SK, Punnam SR, Verma G, Ruberg FL. Persistent left superior vena cava: a case report and review of literature. Cardiovasc Ultrasound 2008;6:50.ArticlePubMedPMCPDF
- 3. Abdel Razek AA, Al-Marsafawy H, Elmansy M, El-Latif MA, Sobh D. Computed tomography angiography and magnetic resonance angiography of congenital anomalies of pulmonary veins. J Comput Assist Tomogr 2019;43:399–405.ArticlePubMed
- 4. Kawsara A, Núñez Gil IJ, Alqahtani F, Moreland J, Rihal CS, Alkhouli M. Management of coronary artery aneurysms. JACC Cardiovasc Interv 2018;11:1211–23.ArticlePubMed
- 5. Pham V, Hemptinne Q, Grinda JM, Duboc D, Varenne O, Picard F. Giant coronary aneurysms, from diagnosis to treatment: a literature review. Arch Cardiovasc Dis 2020;113:59–69.ArticlePubMed
- 6. Abou Sherif S, Ozden Tok O, Taşköylü Ö, Goktekin O, Kilic ID. Coronary artery aneurysms: a review of the epidemiology, pathophysiology, diagnosis, and treatment. Front Cardiovasc Med 2017;4:24.ArticlePubMedPMC
- 7. Gordon JB, Kahn AM, Burns JC. When children with Kawasaki disease grow up: myocardial and vascular complications in adulthood. J Am Coll Cardiol 2009;54:1911–20.ArticlePubMedPMC
- 8. Thangathurai J, Kalashnikova M, Takahashi M, Shinbane JS. Coronary artery aneurysm in Kawasaki disease: coronary CT angiography through the lens of pathophysiology and differential diagnosis. Radiol Cardiothorac Imaging 2021;3:e200550.ArticlePubMedPMC
- 9. Al-Hijji M, El Sabbagh A, El Hajj S, AlKhouli M, El Sabawi B, Cabalka A, et al. Coronary artery fistulas: indications, techniques, outcomes, and complications of transcatheter fistula closure. JACC Cardiovasc Interv 2021;14:1393–406.ArticlePubMed
References
Figure & Data
References
Citations


 E-Submission
E-Submission Yeungnam University College of Medicine
Yeungnam University College of Medicine PubReader
PubReader ePub Link
ePub Link Cite
Cite



