PubMed Central, CAS, DOAJ, KCI

Articles
- Page Path
- HOME > J Yeungnam Med Sci > Volume 38(3); 2021 > Article
-
Original article
Personal experience with microvascular decompression and partial sensory rhizotomy for trigeminal neuralgia -
Jung Hwan Lee
 , Jae Meen Lee
, Jae Meen Lee , Chang Hwa Choi
, Chang Hwa Choi
-
Yeungnam University Journal of Medicine 2021;38(3):202-207.
DOI: https://doi.org/10.12701/yujm.2020.00745
Published online: November 23, 2020
Department of Neurosurgery, Pusan National University Hospital, Busan, Korea
- Corresponding author: Chang Hwa Choi, MD, PhD Department of Neurosurgery, Pusan National University Hospital, 179 Gudeok-ro, Seo-gu, Busan 49241, Korea Tel: +82-51-240-7257 Fax: +82-51-244-8373 E-mail: chwachoi@pusan.ac.kr
Copyright © 2021 Yeungnam University College of Medicine
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 7,749 Views
- 122 Download
- 6 Crossref
Abstract
-
Background
- Trigeminal neuralgia (TN) is a severe, paroxysmal pain in the distribution of the fifth cranial nerve. Microvascular decompression (MVD) is the most widely used surgical treatment for TN. We undertook this study to analyze the effects of and complications of MVD and to refine the surgical procedure for treating TN.
-
Methods
- A total of 88 patients underwent for TN underwent surgery at our hospital. Among them, 77 patients underwent MVD alone, and 11 underwent partial sensory rhizotomy (PSR) with or without MVD. The medical records of these patients were retrospectively analyzed for patient characteristics, clinical results, offending vessels, and complications if any.
-
Results
- The mean follow-up duration was 43.2 months (range, 3–216 months). The most common site of pain was V2+V3 territory (n=27), followed by V2 (n=25) and V3 (n=23). The most common offending vessels were the superior cerebellar artery and anterior inferior cerebellar artery in that order. The overall rate of postoperative complications was 46.1%; however, most complications were transient. There were two cases of permanent partial hearing disturbance. In the MVD alone group, the cure rate was 67.5%, and the improvement rate was 26.0%. Among 11 patients who underwent PSR with or without MVD, the cure rate was 50.0%, and the improvement rate was 30.0%.
-
Conclusion
- The clinical results of MVD were satisfactory. Although the outcomes of PSR were not as favorable as those of pure MVD in this study, PSR can be considered in cases where there is no significant vascular compressive lesion or uncertainty of the causative vessel at the surgery.
- Trigeminal neuralgia (TN) is an intermittent, severe, and electric shock-like facial pain [1,2]. This pain is not life-threatening, but it can seriously deteriorate the quality of life [2]. Various treatment modalities have been used to treat TN, including pharmacotherapy, stereotactic radiosurgery, suboccipital craniotomy with microvascular decompression (MVD) or partial sensory rhizotomy (PSR), and percutaneous procedures such as glycerol rhizotomy, balloon microcompression or radiofrequency rhizotomy [1,2].
- The exact pathogenesis of TN remains unclear; however, it is well-known that vascular compression at the root entry zone (REZ) of the trigeminal nerve is one of the major causes of TN [3]. Thus, MVD has been widely applied to treat TN, and many positive results have been reported [4,5]. Since PSR was first described by Frazier [6], there have been various modifications in the surgical technique [7]. PSR is performed either alone or in combination with MVD, and relatively good results have been reported [3,7].
- In this study, we describe our experience with suboccipital craniotomy in patients with TN and present a thorough review of the literature.
Introduction
- 1. Patient population
- We retrospectively reviewed the medical records and radiologic findings of patients with TN who underwent suboccipital craniotomy with MVD or PSR from 1994 to 2013. Surgeries were performed in patients with typical TN who did not respond to other treatments. All patients underwent preoperative magnetic resonance (MR) imaging using three-dimensional Fourier transformation constructive interference in steady state (3DFT-CISS). Eighty-eight patients underwent 89 surgeries because one patient underwent reoperation. The follow-up period ranged from 3 to 216 months, with an average of 43.2 months.
- 2. Surgical technique
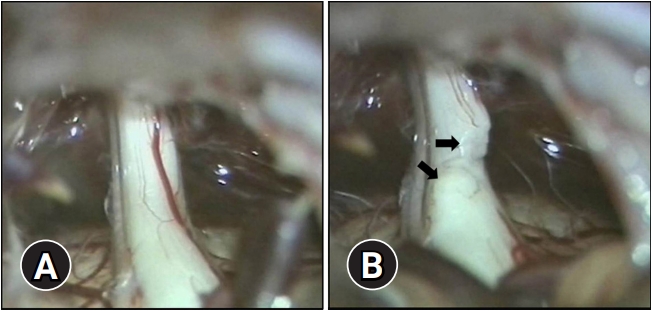
- All surgeries were performed by a senior neurosurgeon. Under general endotracheal anesthesia, patients were placed in the lateral decubitus position. Intraoperative monitoring was performed. After head fixation, a curvilinear skin incision of approximately 7 to 8 cm was made behind the hairline at the posterior of the mastoid process. Then, a retromastoid craniectomy of approximately 3 to 4 cm was performed. The craniectomy was extended to the border of the transverse sinus and sigmoid sinus. The exposed dura was incised in an inverted ‘T’ shape toward the junction of the transverse and sigmoid sinuses. Each incised dura was fixed superiorly (to the occipital bone) and laterally (to the mastoid process). After the cerebellum was gently retracted, the cerebrospinal fluid was drained. The arachnoid membrane was dissected sufficiently to expose the space between the tentorium of the cerebellum and the facial-auditory nerve complex. The trigeminal nerve and the REZ were then found. In case a compressive vessel was observed near the REZ, it was carefully detached from the latter, and horseshoe- or stick-shaped polytetrafluoroethylene sponges were inserted between the two. If there was no compressive vessel, PSR was performed. We cut the sensory root to be adjacent to the pons. The root was cut about 50% to 70% from the caudolateral side (Fig. 1).
- 3. Pain assessment
- We evaluated the surgical outcomes in patients using the Barrow Neurological Institute (BNI) pain scale (Table 1) [8]. The categories of pain relief evaluated by this scale included cure (BNI pain score I or II), improved (BNI score III), and poor (BNI score IV or V). The postoperative complications were also reviewed.
- 4. Radiographic and intraoperative finding analysis
- We compared the MR findings with the intraoperative findings and analyzed the treatment results according to the intraoperative degree of compression of the causative vessel. The surgical techniques were either MVD, PSR, or MVD with PSR, and we examined the outcomes of each.
- 5. Statistical analysis
- All statistical analyses were conducted using IBM SPSS ver. 22.0 (IBM Corp., Armonk, NY, USA). Categorical data were analyzed using the chi-square test or Fisher exact test. The p-values of less than 0.05 were considered statistically significant.
Materials and methods
- 1. Patient characteristics
- The study population included 54 females and 34 males. The mean age of patients was 56.9 years (range, 29–82 years). The distribution and duration of the pain, and the previous treatment modalities are summarized in Table 2.
- 2. Intraoperative findings
- Of the 88 patients who underwent surgery, we could not find the offending vessel in five cases. In the rest of the patients, the superior cerebellar artery was the most common offending vessel. The offending vessels are summarized in Table 3.
- In most cases (79 cases), the suspected offending vessel in 3DFT-CISS images was consistent with the intraoperative findings. There were three cases in which there was no offending vessel in either 3DFT-CISS images or intraoperative findings. In two cases, vessels were suspected as causative in 3DFT-CISS images, but could not be found during surgery. Inversely, in four cases, no offending vessels were observed in 3DFT-CISS images, but we identified them intraoperatively.
- We performed PSR in five patients with no offending vessel identified. The rest of the patients were divided into two groups according to the degree of nerve compression; the compression group and the contact group. The compression group (n=59) consisted of patients with nerve deformation due to vascular compression. On the contrary, patients, in whom there was a mere neurovascular contact without any nerve deformation, were classified as the contact group (n=24). Patients in the compression group underwent MVD. Patients in the contact group also underwent MVD; however, six patients, in whom the causative vessel was small and could not be identified as the definite cause, underwent MVD combined with PSR.
- 3. Relationship between outcomes and surgical modalities
- The preoperative BNI scores of the patients who underwent MVD alone (n=77) were IV in eight patients and V in 69 patients. The postoperative BNI scores of these patients were I in 33 patients and II in 20 patients. Therefore, the cure rate with MVD alone was 68.8%. The preoperative BNI scores of the patients who underwent PSR with or without MVD (n=11) were IV in three patients and V in eight patients, and their postoperative BNI scores were I in six patients. Thus, the cure rate with PSR with or without MVD was 54.5% (Table 4). When the outcomes of the MVD alone group and PSR with or without MVD group were analyzed statistically, there was no significant difference (p=0.444). All patients with improved pain after surgery discontinued medication; however, patients who had sustained pain after surgery (postoperative BNI scores IV and V) continued medication for pain relief.
- 4. Complications and pain recurrence
- We observed postoperative complications in 41 patients, and dizziness was the most common. Almost all complications were temporary, but permanent partial hearing disturbance was also observed in two cases. The complications are summarized in Table 5. There were three cases in which symptoms remained after the surgery and 10 cases in which pain recurred. Pain recurrence was observed in less than a month in two cases, within a year in five cases, and within 5 years in three cases. Among the patients with recurrence of pain, four patients underwent gamma-knife radiosurgery, and the rest resumed medication. Persistent symptoms were identified only in patients who underwent PSR. Among the patients with pain recurrence, eight belonged to the contact group and two belonged to the compression group. One patient who had recurrence underwent reoperation, and PSR was performed because there was no significant compression or adhesion in the operative field.
Results
- Since Dandy [9] first introduced MVD for TN, it was thought to be the most reasonable method of treating TN [10]. The outcomes of MVD for TN have been quite good even in recent reports from the last 10 years [11-13]. In our series, the cure rate for MVD was 68.8%, and the efficacy rate (cure+improvement) was 87%. In this study, we observed pain recurrence in 13% of patients, and one patient underwent reoperation. The results of our series of MVD for TN are similar to those of previous studies.
- Although MVD is an effective surgery for treating TN, the absence of neurovascular compression in TN is also well-known. Some authors have reported that an offending vessel is absent in nearly 20% of patients with TN [14,15]. In some cases, neurovascular compression is not confirmed by MR images. There are various methods to treat this type of TN, including stereotactic radiosurgery, radiofrequency thermal rhizotomy, balloon compression, and retrogasserian glycerol rhizotomy [16]. On the contrary, in some cases, preoperative MR images suggest that there is a causative vessel, but it is not present in the actual surgical field. In our series, there were two cases in which we suspected neurovascular compression in preoperative MR images, but did not find it intraoperatively. If no causative vessel is identified while performing suboccipital craniotomy for MVD, PSR is widely used.
- PSR is performed instead of MVD in the absence of neurovascular compression, and it may be performed simultaneously with MVD [17]. A literature review of recent studies on PSR showed relatively good results [5,18,19]. Toda [17] reported that excellent results (pain-free without medication) of PSR ranged from 48% to 86%. In our series, the cure rate was 54.5%, and the efficacy rate was 72.7%. Pain recurred in one case. The results of our series for PSR are similar to those of previous studies.
- In addition to PSR, ‘nerve combing’ has been introduced as another surgical option that can be performed if there is no causative vessel after suboccipital craniotomy [16]. This operation has been called as ‘neurocombing,’ ‘nerve brushing,’ and ‘internal neurolysis’ [20,21]. This procedure is performed as follows: after the same procedure as MVD, if there is no causative vessel, the sensory root of the TN is longitudinally split into two to six fascicles near the REZ [16,20]. The pain relief rates with nerve combing have been reported to be 72% to 82.1% [20]. However, the mechanism of pain relief is unclear, and there have been no comparative studies with other surgical techniques (MVD or PSR).
- Generally, serious complications related to suboccipital craniotomy, such as facial palsy, cerebellar hemorrhage, and cerebellar edema, can be avoided by an experienced surgeon; however, mild complications are inevitable [22]. Mild complications include headache, nausea, dizziness, hearing change, and face paresthesia. Headache and dizziness are the most common [22,23]. In our series, dizziness was the most common complication, followed by facial paresthesia. Other complications included cerebrospinal fluid leakage and hearing loss. Most complications were transient, and only two patients experienced permanent partial hearing disturbance. There was no postoperative mortality. Complications in this study correspond with findings from previous literature [23]. Permanent complications mainly occurred in cases where surgeries were performed independently. These complications were presumed to be caused by excessive cerebellar traction due to a lack of experience of surgeons. Various surgical tips are needed to prevent these complications and improve surgical outcomes. For instance, sharp microdissection of the arachnoid membrane and slow suction of cerebrospinal fluid are needed rather than using retracting blades. In addition, it is better to use a narrow suction tip [10].
- In a recent review article, pain recurrence after MVD for TN was reported to be 14.3% [24]. In this study, pain recurrence after MVD for TN was 12.8%, and it was more common in the contact group. The presence of vascular compression is known to be a major factor in the recurrence of pain, and the absence of compression has been shown to be associated with lower rates of recurrence [25,26]. These findings are consistent with our series. Contrastingly, there was no pain recurrence after PSR with or without MVD in our series. However, no pain relief was seen in three cases (27.3%). Generally, the recurrence rate with PSR is reported to be higher than that with MVD [3,5,19]. There was no recurrence in our patients who underwent PSR; however, the short follow-up period and the small number of cases did not allow us to draw any definitive conclusions.
- In this study, the clinical results for MVD were satisfactory; the cure rate was 68.8%. On the contrary, the cure rate with PSR was 54.5%. Although the outcomes of PSR were not as favorable as those of pure MVD in this study, PSR can be considered when there is no significant vascular compressive lesion, or the causative vessel is uncertain intraoperatively.
Discussion
-
Ethical statements
This retrospective study was performed with approval from the Institutional Review Board (IRB) of Pusan National University Hospital (IRB No: 1803-021-065).
-
Conflicts of interest
No potential conflicts of interest relevant to this article were reported.
-
Funding
This work was supported by a 2-year Research Grant from Pusan National University.
-
Author contributions
Conceptualization: CHC, JHL; Data curation: all authors; Formal analysis, Project administration, Supervision: CHC; Investigation: JHL; Writing-original draft: JML; Writing-review & editing: all authors.
Notes
| Offending vessel | No. |
|---|---|
| Superior cerebellar artery | 49 |
| Anterior inferior cerebellar artery | 18 |
| Vertebral artery | 4 |
| Basilar artery | 2 |
| Posterior inferior cerebellar artery | 2 |
| Posterior cerebral artery | 1 |
| Superior petrosal vein | 4 |
| Multiplea) | 3 |
| Absent | 5 |
| Total | 88 |
- 1. Kang IH, Park BJ, Park CK, Malla HP, Lee SH, Rhee BA. A clinical analysis of secondary surgery in trigeminal neuralgia patients who failed prior treatment. J Korean Neurosurg Soc 2016;59:637–42.ArticlePubMedPMCPDF
- 2. Zhang L, Zhang Y, Li C, Zhu S. Surgical treatment of primary trigeminal neuralgia: comparison of the effectiveness between MVD and MVD+PSR in a series of 210 patients. Turk Neurosurg 2012;22:32–8.ArticlePubMed
- 3. Gao J, Fu Y, Guo SK, Li B, Xu ZX. Efficacy and prognostic value of partial sensory rhizotomy and microvascular decompression for primary trigeminal neuralgia: a comparative study. Med Sci Monit 2017;23:2284–91.ArticlePubMedPMC
- 4. Klun B. Microvascular decompression and partial sensory rhizotomy in the treatment of trigeminal neuralgia: personal experience with 220 patients. Neurosurgery 1992;30:49–52.ArticlePubMed
- 5. Bederson JB, Wilson CB. Evaluation of microvascular decompression and partial sensory rhizotomy in 252 cases of trigeminal neuralgia. J Neurosurg 1989;71:359–67.ArticlePubMed
- 6. Frazier CH. Trigeminal neuralgia: fourteen years experience with fractional section of the sensory root as the major operation. JAMA 1927;89:1742–4.Article
- 7. Kim SH, Choi CH. The efficacy of microvascular decompression for trigeminal neuralgia. J Korean Neurosurg Soc 2005;37:357–63.
- 8. Rogers CL, Shetter AG, Fiedler JA, Smith KA, Han PP, Speiser BL. Gamma knife radiosurgery for trigeminal neuralgia: the initial experience of The Barrow Neurological Institute. Int J Radiat Oncol Biol Phys 2000;47:1013–9.ArticlePubMed
- 9. Dandy WE. Concerning the cause of trigeminal neuralgia. AM J Surg 1934;24:447–55.Article
- 10. Xia L, Zhong J, Zhu J, Wang YN, Dou NN, Liu MX, et al. Effectiveness and safety of microvascular decompression surgery for treatment of trigeminal neuralgia: a systematic review. J Craniofac Surg 2014;25:1413–7.ArticlePubMed
- 11. Bond AE, Zada G, Gonzalez AA, Hansen C, Giannotta SL. Operative strategies for minimizing hearing loss and other major complications associated with microvascular decompression for trigeminal neuralgia. World Neurosurg 2010;74:172–7.ArticlePubMed
- 12. Ferroli P, Acerbi F, Tomei M, Tringali G, Franzini A, Broggi G. Advanced age as a contraindication to microvascular decompression for drug-resistant trigeminal neuralgia: evidence of prejudice? Neurol Sci 2010;31:23–8.ArticlePubMedPDF
- 13. Günther T, Gerganov VM, Stieglitz L, Ludemann W, Samii A, Samii M. Microvascular decompression for trigeminal neuralgia in the elderly: long-term treatment outcome and comparison with younger patients. Neurosurgery 2009;65:477–82.ArticlePubMedPDF
- 14. Lee A, McCartney S, Burbidge C, Raslan AM, Burchiel KJ. Trigeminal neuralgia occurs and recurs in the absence of neurovascular compression. J Neurosurg 2014;120:1048–54.ArticlePubMed
- 15. Ishikawa M, Nishi S, Aoki T, Takase T, Wada E, Ohwaki H, et al. Operative findings in cases of trigeminal neuralgia without vascular compression: proposal of a different mechanism. J Clin Neurosci 2002;9:200–4.ArticlePubMed
- 16. Zhao H, Zhang X, Tang D, Li S. Nerve combing for trigeminal neuralgia without vascular compression. J Craniofac Surg 2017;28:e15–6.ArticlePubMed
- 17. Toda K. Operative treatment of trigeminal neuralgia: review of current techniques. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2008;106:788–805. e1-6.ArticlePubMed
- 18. Abhinav K, Love S, Kalantzis G, Coakham HB, Patel NK. Clinicopathological review of patients with and without multiple sclerosis treated by partial sensory rhizotomy for medically refractory trigeminal neuralgia: a 12-year retrospective study. Clin Neurol Neurosurg 2012;114:361–5.ArticlePubMed
- 19. Koopman JS, de Vries LM, Dieleman JP, Huygen FJ, Stricker BH, Sturkenboom MC. A nationwide study of three invasive treatments for trigeminal neuralgia. Pain 2011;152:507–13.ArticlePubMed
- 20. Liang X, Dong X, Zhao S, Ying X, Du Y, Yu W. A retrospective study of neurocombing for the treatment of trigeminal neuralgia without neurovascular compression. Ir J Med Sci 2017;186:1033–9.ArticlePubMedPDF
- 21. Ko AL, Ozpinar A, Lee A, Raslan AM, McCartney S, Burchiel KJ. Long-term efficacy and safety of internal neurolysis for trigeminal neuralgia without neurovascular compression. J Neurosurg 2015;122:1048–57.ArticlePubMed
- 22. Zhong J, Zhu J, Sun H, Dou NN, Wang YN, Ying TT, et al. Microvascular decompression surgery: surgical principles and technical nuances based on 4000 cases. Neurol Res 2014;36:882–93.ArticlePubMed
- 23. Oesman C, Mooij JJ. Long-term follow-up of microvascular decompression for trigeminal neuralgia. Skull Base 2011;21:313–22.ArticlePubMedPMCPDF
- 24. Phan K, Rao PJ, Dexter M. Microvascular decompression for elderly patients with trigeminal neuralgia. J Clin Neurosci 2016;29:7–14.ArticlePubMed
- 25. Theodros D, Rory Goodwin C, Bender MT, Zhou X, Garzon-Muvdi T, De la Garza-Ramos R, et al. Efficacy of primary microvascular decompression versus subsequent microvascular decompression for trigeminal neuralgia. J Neurosurg 2017;126:1691–7.ArticlePubMed
- 26. Mendoza N, Illingworth RD. Trigeminal neuralgia treated by microvascular decompression: a long-term follow-up study. Br J Neurosurg 1995;9:13–9.Article
References
Figure & Data
References
Citations

- Revisiting the Efficacy of Redo Microvascular Decompression for Trigeminal Neuralgia
Zhongding Zhang, Hua Zhao, Yinda Tang, Baimiao Wang, Qing Yuan, Ying Zhang, Yihua Li, Jun Zhong, Shiting Li
World Neurosurgery.2024; 186: e335. CrossRef - A systematic review on the efficacy of adjunctive surgical strategies during microvascular decompression for trigeminal neuralgia without intraoperative evidence of neurovascular conflict
Nicola Montano, Grazia Menna, Alessandra Musarra, Renata Martinelli, Alessandro Izzo, Quintino Giorgio D’Alessandris, Manuela D’Ercole, Alessandro Olivi
Neurosurgical Review.2024;[Epub] CrossRef - Progress in Surgical Treatment of Trigeminal Neuralgia
滨 何
Advances in Clinical Medicine.2023; 13(02): 2313. CrossRef - How Far Has Radiofrequency Thermocoagulation Come Along as a Treatment Procedure in Treating Trigeminal Neuralgia Patients?
Stephen D Howard, Varun Soti
Cureus.2023;[Epub] CrossRef - Historical aspects of the problem of treatment of trigeminal neuralgia and the role of neurosurgical methods in its solution (literature review)
A. N. Zhurkin, A. V. Semenov, V. A. Sorokovikov, N. V. Bartul
Acta Biomedica Scientifica.2021; 6(4): 123. CrossRef - Trigeminal Neuralgia: Current Approaches and Emerging Interventions
Risheng Xu, Michael E Xie, Christopher M Jackson
Journal of Pain Research.2021; Volume 14: 3437. CrossRef

 E-Submission
E-Submission Yeungnam University College of Medicine
Yeungnam University College of Medicine PubReader
PubReader ePub Link
ePub Link Cite
Cite


