Clinical significance of exosomal noncoding RNAs in hepatocellular carcinoma: a narrative review
Article information
Abstract
Hepatocellular carcinoma (HCC) is one of the most lethal malignancies worldwide, with poor prognosis owing to its high frequency of recurrence and metastasis. Moreover, most patients are diagnosed at an advanced stage owing to a lack of early detection markers. Exosomes, which are characterized by their cargos of stable intracellular messengers, such as DNA, RNA, proteins, and lipids, play a crucial role in regulating cell differentiation and HCC development. Recently, exosomal noncoding RNAs (ncRNAs), including microRNAs, long ncRNAs, and circular RNAs, have become increasingly important diagnostic, prognostic, and predictive markers of HCC. Herein, we discuss the clinical implications of exosomal ncRNAs, specifically those within the HCC regulatory network.
Introduction
Hepatocellular carcinoma (HCC) is a significant health issue for patients with chronic liver disease, has a high socioeconomic burden, and is the third leading cause of cancer-related mortality worldwide [1,2]. Patients diagnosed with HCC in the early stage, accounting for 30% to 40% of all HCC cases, have a favorable prognosis because of the feasibility of curative treatments, which include liver transplantation, surgical resection, and local ablation therapy [3]. However, most patients with HCC diagnosed at intermediate or advanced stages have a poor prognosis due to high rates of recurrence and metastasis, as well as the limitations of curative treatments [3-6].
In general, current diagnostic methods for detecting HCC rely on clinical radiological modalities, such as computed tomography and magnetic resonance imaging, without the need for histological confirmation because of the potential of liver biopsies to cause serious complications, including bleeding, cancer seeding, rupture, and even death [7,8]. Nonetheless, the sensitivity of radiological modalities for early detection of HCC varies based on the type of modality and diagnostic criteria applied [9,10]. As a serologic diagnostic marker, α‐fetoprotein (AFP) is considered a novel biomarker for early identification of patients with HCC. However, the sensitivity of AFP for detecting HCC is low at 20% to 65% [11].
Recent studies have indicated that exosomal noncoding RNAs (ncRNAs) are potential diagnostic and prognostic markers in patients with HCC. Previously, ncRNAs were considered nonfunctional genes. However, with the development of RNA identification techniques such as next-generation sequencing, various ncRNAs have been shown to regulate target messenger RNAs (mRNAs) and modulate gene expression [12,13]. Recently, dysregulation of ncRNAs has been linked to tumor progression and metastasis in patients with HCC, and ncRNAs are now considered to be promising diagnostic and therapeutic targets for HCC [14].
Based on their nucleotide length and structure, ncRNAs are classified into microRNAs (miRNAs), long ncRNAs (lncRNAs), and circular RNAs (circRNAs). MiRNAs are single-stranded ncRNAs of 19–25 nucleotides that are closely associated with various biological processes and regulate protein expression by mRNA degradation and translational inhibition by binding to the 3′-untranslated region of target pre-mRNAs [15,16]. Given that miRNAs can function as either tumor suppressors or oncogenes, their aberrant expression has been linked to carcinogenesis, including tumor angiogenesis, cell proliferation, tumor invasion, and metastasis [17-20]. LncRNAs, which are ncRNAs exceeding 200 nucleotides in length and typically do not code for proteins, are known for their heterogeneity [21]. Previous studies have indicated that lncRNAs regulate gene expression by acting as miRNA sponges or competing endogenous RNAs (ceRNAs) [22,23]. Their dysregulation has been found to affect carcinogenic processes, including cell differentiation and proliferation, by altering gene expression [24,25]. CircRNAs are endogenous circular ncRNAs with covalently closed linked ends (in contrast to linear RNAs such as miRNAs and lncRNAs) and are produced by the backsplicing of exons and/or introns on precursor mRNAs [26]. Unlike linear RNAs, circRNAs are resistant to RNase because they lack a 5′ cap and 3′ polyadenylated tail [27]. Similar to lncRNAs, the precise roles of circRNAs remain unclear. However, circRNAs has been reported to function as miRNA sponges or ceRNAs at both transcriptional and posttranscriptional levels [28].
Exosomes are nanoscale cup-shaped or double concave disc-shaped vesicles (30–100 nm) secreted by most cell types; they can be detected in body fluids, such as serum, urine, ascitic fluid, and breast milk [29-33]. Exosomes play a crucial role in cell-to-cell communication between donor and recipient cells by fusing with cell receptors and transferring genetic information [34]. Exosomes contain cell-specific mRNAs, ncRNAs (including miRNAs, lncRNAs, and circRNAs), and proteins, which are stabilized in the circulation because of their exosomal protective and antidegradative functions against RNase [35-38] (Fig. 1). Recent study have demonstrated that exosomal miRNAs may serve as potential diagnostic and prognostic markers in patients with malignancies [39].
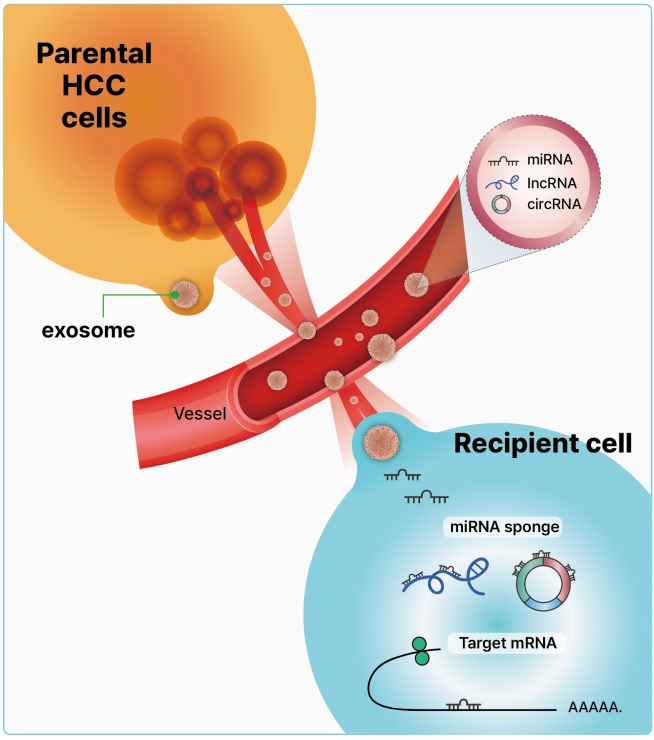
Exosomal noncoding RNAs are secreted via exocytosis or through a blood vessel from parental hepatocellular carcinoma (HCC) cells. These exosomes contain microRNAs (miRNAs), long noncoding RNAs (lncRNAs), and circular RNAs (circRNAs), and fuse with the recipient cell membrane, releasing the exosomal content into the recipient cell. The exosomal noncoding RNAs derived from cancer contribute to intercellular communication, resulting in alterations of genetic information in recipient target messenger RNAs that promote cancer-associated changes.
Given the clinical importance of early detection for improving prognosis and facilitating curative treatment of HCC, the exploration of emerging diagnostic and prognostic biomarkers is warranted [40]. Although exosomal ncRNAs are promising novel biomarkers of HCC, their mechanisms of action in patients with HCC remain unclear. In this review, we describe the clinical applications of HCC-derived exosomal miRNAs, lncRNAs, and circRNAs over the past decade and classify them into diagnostic, prognostic, and therapeutic categories.
Diagnostic markers of the exosomal noncoding RNAs of hepatocellular carcinoma
Early detection of HCC can improve curative treatment options and long-term survival rates. Traditional diagnostic methods for HCC have limited sensitivity, making early detection challenging. Currently, a significant amount of research is focused on liquid biopsy techniques as new diagnostic methods. Recent studies have shown promising results regarding the use of exosomal ncRNAs as effective diagnostic markers in patients with HCC. Through the advancement of purification and isolation techniques for miRNAs using RNA extraction kits, exosomal miRNAs have been specifically identified as diagnostic marker, when compared to exosomal lncRNAs and circRNAs. Table 1 [41-68] summarizes the diagnostic markers of exosomal ncRNAs in patients with HCC.
1. Exosomal microRNAs as diagnostic biomarkers
Previous research has identified elevated levels of plasma miRNA-21 even in the early stages of HCC. Further analysis revealed that the level of plasma miRNA-21 was significantly higher in patients with HCC than in those with chronic hepatitis or in the control group. This study is the first to demonstrate the feasibility of using plasma miRNA-21 as an early diagnostic marker for HCC [69]. Wang et al. [41] demonstrated that exosomal miRNA-21 is a potential diagnostic marker for early HCC. Their study found that serum exosomal miRNA-21 expression was higher in patients with HCC than in healthy individuals and in patients with chronic hepatitis B (CHB). Additionally, exosomal miRNA-21 expression levels were found to be higher than those in exosome-depleted serum and whole serum [41]. However, this study did not identify the target genes associated with HCC.
Owing to the low sensitivity of AFP in the diagnosis of HCC, it has been proposed that the combination of an emerging exosomal miRNA with AFP can enhance the accuracy of HCC diagnosis. Liu et al. [42] identified promising diagnostic biomarkers for non–virus-infected HCC in diethylnitrosamine-induced HCC rats. Specifically, four selected miRNAs (miRNA-10b and miRNA-21 as oncogenes and miRNA-122 and miRNA-200a as tumor suppressor genes) were identified. The combination of exosomal miRNAs and AFP exhibited a more significant diagnostic potential in predicting HCC as compared to the use of AFP alone [42]. Wang et al. [43] found that the serum exosomal levels of miRNA-122, miRNA-148a, and miRNA-1246 were significantly higher in individuals with HCC than in those with liver cirrhosis (LC) or in normal controls (NC). A combination of serum exosomal miRNA-122, miRNA-148a, and AFP levels can be used to discriminate between early HCC and LC. While plasma miRNA-21 is not exosome-derived, a previous study demonstrated that the combination of plasma miRNA-21 and AFP has stronger discriminant power for detecting early HCC than AFP alone [69].
In a domestic study, Sohn et al. [44] found that the use of variable serum exosomal miRNAs resulted in improved diagnostic accuracy for distinguishing HCC from CHB or LC when compared to serum circulating miRNAs. In that study, researchers observed higher serum levels of exosomal miRNA-18a, miRNA-221, miRNA-222, and miRNA-224 and lower levels of exosomal miRNA-101, miRNA-106b, miRNA-122, and miRNA-195 in patients with HCC than in those with CHB or LC. Compared to previous studies, exosomal miRNA expression was similar to that of tissue miRNAs. In HCC tissues, miRNA-18a, miRNA-221, miRNA-222, and miRNA-224 were overexpressed, whereas miRNA-101, miRNA-195, and miRNA-122a were downregulated [70-72]. However, the target genes of these miRNAs in HCC were not identified in the study. Interestingly, serum circulating miRNAs were not significantly different between the HCC and CHB groups, unlike serum exosomal miRNAs. This study suggests that serum exosomal miRNAs are more effective markers than circulating serum miRNAs for differentiating HCC from CHB or LC [44].
Cho et al. [45] found that serum exosomal miRNA-10b-5p is a potential biomarker for early-stage HCC, with an area under the curve (AUC) of 0.934. Han et al. [46] found that miRNA-148a expression in the plasma was significantly decreased in individuals with HCC. When comparing patients with HCC to those with LC, the AUC for plasma miRNA-148a was 0.919 (sensitivity, 89.5%; specificity, 89.5%). In patients with HCC and low or negative AFP, the AUC for plasma miRNA-148a was 0.949 (sensitivity, 90.6%; specificity, 92.6%). MiRNA-148a could be a feasible noninvasive biomarker that may work in conjunction with AFP to diagnose HCC. Fu et al. [47] demonstrated the crucial role of phosphatase and tensin homolog (PTEN) and miRNA-155-5p in aggressive HCC both in vitro and in vivo. In a diethylnitrosamine/N-nitrosomorpholine-induced HCC rat model, miRNA-155-5p overexpression occurred simultaneously with PTEN mRNA suppression. These findings have also been observed in human HCC tissues and cell lines. Therefore, miRNA-155-5p may serve as a novel diagnostic marker and therapeutic target.
2. Exosomal long noncoding RNAs and circular RNAs as diagnostic biomarkers
Recently, research has been conducted to identify not only the basic functions of miRNAs, but also lncRNAs and circRNAs, which can act as miRNA sponges and regulate target genes in patients with HCC. Specifically, circRNAs are resistant to exonuclease degradation because of their covalently closed linked ends, leading to their accumulation in cells and slow degradation [73]. Furthermore, exosomes can protect lncRNAs from degradation by RNase, allowing them to remain stable within exosomes [29]. However, compared with investigations of exosomal miRNAs, those of exosomal lncRNAs and circRNAs are rare in patients with HCC. Table 1 lists the diagnostic biomarkers of the exosomal lncRNAs and circRNAs identified in HCC.
Zhang et al. [48] demonstrated that patients with hepatitis C virus-related HCC have elevated expression levels of lncRNA-HEIH in serum and exosomes, indicating its potential as a biomarker for the diagnosis of HCC. However, the ratio of lncRNA-HEIH in serum versus exosomes is lower in patients with HCC than in those with chronic hepatitis C (CHC). The authors hypothesized that the exosomal lncRNA has limited potential to regulate gene expression until its release from exosomes into the serum. Xie et al. [49] demonstrated a difference in the expression of lncRNA-highly upregulated in liver cancer (HULC) between the tissues and plasma of patients with HCC and healthy controls. The AUC of lncRNA-HULC in predicting HCC was 0.86. Moreover, these data suggest that lncRNA-HULC is associated with malignancy and tumor stage. These results show that lncRNA-HULC expression in the plasma may be used as a noninvasive, innovative biomarker that shows promise for the diagnosis and prognosis of HCC. Wang et al. [50] revealed that miRNA-186 is sequestered by lncRNA-HULC, increasing high-mobility group AT-hook 2 expression, thereby promoting the progression of liver cancer. Ji et al. [51] showed that linc00152 may contribute to the oncogenesis of HCC and may eventually serve as a potential indicator of clinical outcomes by stimulating the mammalian target of rapamycin signaling pathway. Additionally, Kim et al. [52] demonstrated that serum extracellular vesicle-derived linc00853, which was shown to have a sensitivity of 93.75% and specificity of 89.77%, may be a breakthrough promising early HCC surveillance biomarker. Sun et al. [53] reported that lncRNA-00161 levels were higher in patients with HCC than in controls. The expression of lncRNA-00161 was detected in serum exosomes, exosome-free serum, and urine samples, whereas only serum exosomal lncRNA-00161 was overexpressed in patients with HCC as compared to expression in control patients. These findings indicate that circulating serum exosomal lncRNA-00161 may serve as a biomarker for HCC. However, the target genes and associated miRNAs in patients with HCC were not elucidated in this study.
Similar to studies on exosomal miRNA, Xu et al. [54] conducted research on the diagnostic value of combining lncRNA and AFP for HCC. They demonstrated that a combination of serum exosomal lncRNA-ENSG00000258332.1, lncRNA-00635, and AFP may be a potential diagnostic marker for HCC. The AUC for the combination of the two lncRNAs and serum AFP was 0.894, while lncRNA-ENSG00000258332.1 and lncRNA-00635 showed AUCs of 0.719 and 0.750, respectively.
As the regulatory functions of lncRNAs as miRNA sponges or ceRNAs have been elucidated, several studies have recently examined the target miRNAs and genes associated with lncRNAs. Matboli et al. [55] found that serum exosomal lncRNA-RP11-583F2.2 levels were higher in patients with HCC than in those with CHC and in the normal group. The study also found a negative correlation between lncRNA-RP11-583F2.2 and miRNA-1298, which is believed to act as a sponge for miRNA-1298. Furthermore, lncRNA-RP11-583F2.2 demonstrated higher sensitivity and specificity than AFP (sensitivity, 98.3% vs. 90%; specificity, 91.7% vs. 85%). However, the authors did not discuss the putative mechanisms of the target genes. Gao et al. [56] showed that tumor size, TNM stage, and AFP levels were associated with upregulated plasma SNHG1 expression levels. According to receiver operating characteristic analysis, SNHG1 had a great diagnostic performance (AUC, 0.86–0.97) for distinguishing between patients with HCC and healthy control individuals. Li et al. [74] illustrated the mechanism of sorafenib resistance using lncRN-SNHG1. miR-21, whose nuclear translocation is triggered by sorafenib, promotes the nuclear expression of lncRNA-SNHG1, which in turn leads to sorafenib resistance by activating the Akt pathway. These findings indicate that SNHG1 could be a beneficial target for the treatment of patients with HCC that has developed sorafenib resistance.
Li et al. [57] found significant upregulation of serum exosomal lncRNA-FAL1 in patients with HCC. Regarding its oncogenic role, lncRNA-FAL1 has been reported to promote cell proliferation, invasion, and epithelial-mesenchymal transition (EMT) by increasing the expression of AFP and zinc finger E-box-binding homeobox 1 (ZEB1). Exosomal lncRNA-FAL1 enhances cancer cell proliferation and metastasis by acting as a miR-1236 sponge. In a previous study, miRNA-1236 was found to downregulate HCC proliferation, migration, and invasion as a tumor suppressor, inhibiting the phosphoinositide 3-kinase (PI3K)/Akt pathway through elevated PTEN expression via the repression of AFP [75]. Wang et al. [58] showed that lncRNA-UCA1 promotes the malignant development of human HCC and identified a new regulatory cascade for the UCA1-miRNA-216b-FGFR1-extracellular signal-regulated kinase (ERK) signaling pathway in HCC. These findings are relevant for potential HCC diagnostic markers and/or therapies. Hou et al. [59] revealed that lncRNA-metastasis-associated lung adenocarcinoma transcript 1 (MALAT1) stimulates HCC migration and invasion via two different mechanisms: it sponges miRNA-204 and releases NAD-dependent deacetylase sirtuin-1 (SIRT1) from miRNA-204. Furthermore, through its interaction with miRNA-200a in hypoxic Hep3B cells, MALAT1 is engaged in proliferation, migration, invasion, and apoptosis, revealing a novel mechanism of its involvement in the development of hypoxic HCC [60]. According to Cao et al. [61], lncRNA-UBE2CP3 induces EMT, which in turn enhances HCC metastasis. Additionally, they demonstrated that patients with HCC had elevated blood levels of lncRNA-UBE2CP3. This study determined the significance of lncRNA-UBE2CP3 in predicting HCC development and clinical outcomes. Based on these findings, lncRNA-UBE2CP3 may serve as a potential diagnostic biomarker. Jing et al. [62] showed that lncRNA-SPRY4-IT1 is a key player in the carcinogenesis of HCC and that SPRY4-IT1 might be used as a diagnostic marker for the disease. Nevertheless, these reports did not describe the target genes and related miRNAs in patients with HCC. Huang et al. [63] compared the diagnostic performance of lncRNAs such as HULC, linc00152, UCA1, MALAT1, UBE2CP3, and SPRY4-IT1 using AUC values, and the respective AUC values were 0.796 (95% confidence interval [CI], 0.734–0.858), 0.895 (95% CI, 0.854–0.936), 0.858 (95% CI, 0.810–0.907), 0.768 (95% CI, 0.706–0.830), 0.812 (95% CI, 0.754–0.866), and 0.808 (95% CI, 0.750–0.866), respectively.
MiRNA-324-5p overexpression serves as a tumor suppressor that decreases migration and invasion in HCC by regulating the expression of matrix metalloproteinase (MMP) 2, MMP9, erythroblast transformation-specific (ETS) 1, and specificity protein 1 [76]. Huang et al. [64] reported that plasma exosomal lncRNA-85 functions as an oncogene and potentially acts as a sponge for miRNA-324-5p, thereby playing a significant role in HCC tumorigenesis. This suggests that lncRNA-85 is a potential biomarker for HCC. Interestingly, the high expression level of exosomal lncRNA-85 is a distinguishing marker for patients with AFP-negative HCC compared to NC and patients with LC, with an AUC value of 0.869 [64].
There is one report on the use of exosomal circRNAs as diagnostic markers in patients with HCC. Chen et al. [65] illustrated that elevated levels of plasma exosomal circ-0051443 serve as a suitable distinguishing flag for patients with HCC, in contrast to NC, exhibiting an AUC value of 0.8089. Zhang et al. [66] demonstrated that circ_104075 is elevated in HCC and functions as a ceRNA that absorbs miR-582-3p and increases yes-associated protein (YAP) production, thereby stimulating HCC carcinogenesis. Circ_104075 was shown to have a higher sensitivity of 96.0% and specificity of 98.3% for the diagnosis of HCC than other ncRNA biomarkers. Circ _104075 could be a new therapeutic target and diagnostic biomarker for HCC. According to Li et al. [67], circSMARCA5 triggers apoptosis and hinders the growth, invasion, and metastasis of HCC cells. CircSMARCA5 may be a useful biomarker for HCC monitoring and prediction, particularly in patients with AFP levels of <200 ng/mL. Recently, it was reported that a network of noncoding regulatory RNAs, driven by circ‐chromodomain Y like (circ‐CDYL), functioned specifically in the early stages of HCC. The characteristics of liver tumor-initiating cells expressing epithelial cell adhesion molecule (EpCAM) were enhanced by circ-CDYL. Using miRNA-892a and miRNA-328-3p as sponges, circ-CDYL cooperates with mRNAs encoding hepatoma-derived growth factor and hypoxia-inducible factor asparagine hydroxylase. These results reveal a noncoding regulatory RNA system that is circRNA-driven in the early stages of HCC, providing opportunities for early detection and therapy [68].
Prognostic markers of exosomal noncoding RNAs of hepatocellular carcinoma
Several studies have indicated that exosomal ncRNAs are potential prognostic factors in patients with HCC. Our focus is on the clinical implications of exosomal ncRNAs in relation to tumor characteristics such as stage, vessel invasion, metastasis, and target gene expression. Table 2 [41,65,77-93] lists the exosomal ncRNAs known as prognostic markers.
1. Exosomal microRNAs as prognostic markers
In a previous study, high expression of circulating miRNA-21 was linked to PTEN/Akt signaling activation, which promotes EMT and tumor progression, and is associated with poor prognosis in patients with HCC [69]. Wang et al. [41] discovered that high levels of serum exosomal miRNA-21 were positively correlated with LC and advanced tumor stage, indicating that serum exosomal miRNA-21 could be a potential predictive marker for HCC risk. However, this study did not mention the target genes or signaling pathways involved.
The mitogen-activated protein kinase (MAPK)/ERK signaling pathway is considered an antiapoptotic pathway that plays a vital role in the malignant proliferation of tumor cells by regulating apoptosis-related proteins [94,95]. Recent studies have indicated that MAPK/ERK-related molecules are active in both gastric and breast cancers [96,97]. Qu et al. [77] found a positive correlation between serum exosomal miRNA-665 levels and advanced clinical parameters, including tumor size and patient mortality, suggesting the involvement of serum exosomal miRNA-665 in HCC development. Exosomal miRNA-665 promoted HCC cell proliferation by increasing the phosphorylation of ERK, as demonstrated by western blotting and in vivo experiments. Anti-miRNA-665 has also been shown to be effective in suppressing exosomal miRNA-665 expression.
Recently, several studies demonstrated that tumor-derived exosomal miRNAs play critical roles in promoting cancer metastasis by regulating target gene expression. Cancer-associated fibroblasts (CAFs), which are activated fibroblasts, are strongly associated with tumor progression and metastasis, and enhance the tumorigenicity of tumor cells via interleukin-6 and interleukin-8 signaling [98,99]. Fang et al. [78] demonstrated that highly metastatic HCC cells located in the metastatic lung niche secrete exosomal miRNA-1247-3p as an onco-miRNA. This secretion leads to the downregulation of β-1,4-galactosyltransferase III and the activation of CAFs via the beta-1-integrin-nuclear factor-κB signaling pathway. Yang et al. [79] reported that hepatoma-derived exosomal miRNA 92a-3p has the potential to promote cancer progression by inducing EMT, which is a critical step in metastasis, downregulation of the tumor suppressor gene PTEN, and activation of the Akt/Snail pathway. High expression of miRNA-92a-3p, facilitated by transcription factors E2F1 and c-Myc, is associated with cancer progression and lung metastasis. This indicates that exosomal miRNA-92a-3p may serve as a predictive marker of HCC metastasis.
Several studies have examined the association of HCC prognosis with tumor angiogenesis. Previous research has shown that knockdown of SMAD family member 4 (SMAD4) and signal transducer and activator of transcription 6 (STAT6) promotes endothelial cell migration and neovascularization, with SMAD4 and STAT6 potentially serving as inhibitory regulators of angiogenesis [100,101]. Lin et al. [80] demonstrated that exosomal miRNA-210 could be delivered from HCC cells to endothelial cells, thereby enhancing tumor angiogenesis by suppressing the expression of SMAD4 and STAT6. This study is the first to detect communication between HCC and endothelial cells using exosomal miRNA-210.
Attenuation of junction integrity between endothelial cells promotes tumor metastasis. The endothelial cell junction is composed of the proteins vascular endothelial-cadherin (VE-Cad), catenins, and zonula occludens [102]. Fang et al. [81] demonstrated that exosomal miRNA-103 derived from hepatoma cells increases vascular permeability by reducing endothelial junction integrity and inhibiting the expression of VE-Cad, p120-catenin, and zonula occludens, which promotes HCC metastasis. Hepatoma cell-secreted miRNA-103 has the potential to serve as both a predictive marker and therapeutic target for HCC metastasis.
Several exosomal miRNAs that function as tumor suppressors in HCC have been reported. Previous studies have shown that miRNA-125b inhibits the EMT, migration, and invasion of HCC cells by downregulating SMAD2/4, SIRT17, SUV39H1, LIN28B, and phosphatidylinositol glycan anchor biosynthesis class F [103-107]. Liu et al. [82] demonstrated that serum exosomal miRNA-125b is a predictive factor associated with time to recurrence (TTR) and overall survival (OS) in patients with HCC. In that study, it was evident that patients with HCC and low miRNA-125b expression had shorter TTR and OS than those with high miRNA-125b expression. Therefore, downregulation of miRNA-125 may be linked to the growth and invasion of HCC cells. According to Shi et al. [83], low expression of serum exosomal miRNA-638 predicts poor prognosis with poor OS in patients with HCC. A negative association was observed between serum exosomal miRNA-638 and clinicopathological tumor characteristics, such as tumor size, vascular invasion, and TNM stage, in patients with HCC. In a previous study, the downregulation of miRNA-638 in HCC facilitated angiogenesis via regulation of vascular endothelial growth factor (VEGF) [108]. Coulouarn et al. [109] reported that miRNA-122 expression tends to be downregulated in HCC, which was linked to worse prognosis; increased tumor growth, invasion, and metastasis; and poor differentiation. In addition, Chen et al. [84] reported that serum miRNA-34a is downregulated in patients with HCC, suggesting its potential as a promising marker for HCC detection and surveillance. Through experiments using a mirRNA-34a mimic, Dang et al. [110] showed that miRNA-34a acts as a tumor suppressor miRNA in HCC, targeting phospho-ERK1/2, phospho-STAT5 signaling, and c-MET. MiRNA-497 is a prognostic marker of HCC. Yan et al. [85] demonstrated that miRNA-497 inhibits VEGF and astrocyte elevated gene-1 (AEG-1) production, which reduces the angiogenesis and metastasis of HCC cells, both in vitro and in vivo.
2. Exosomal long noncoding RNAs as prognostic markers
LncRNA-ATB is an oncogenic factor that induces EMT, tumor invasion, and metastasis by upregulating ZEB1 and ZEB2 [111]. Lee et al. [86] have shown that serum exosomal lncRNA-ATB is a potential prognostic factor for HCC. Elevated levels of exosomal lncRNA-ATB are linked to aggressive tumor features, including TMN stage and portal vein thrombosis, resulting in reduced OS and progression-free survival. In that study, elevated expression of exosomal miRNA-21 was found to be an independent predictor of assessment, and there were no associations between miRNA-21 and lncRNA-ATB. According to Huang et al. [87], lncRNA-PTTG3P may stimulate PI3K/Akt signaling in HCC and upregulate pituitary tumor-transforming 1, which in turn may promote tumor development and metastasis and could be an effective prognostic indicator of HCC.
Recent studies have investigated the roles of lncRNAs in the treatment of HCC. Although radiofrequency ablation (RFA) is a curative treatment option for patients with early HCC, inadequate RFA has been linked to local recurrence and HCC metastasis [88,112]. According to Ma et al. [88], elevated levels of exosomal lncRNA-ASMTL-AS1 are responsible for the malignant transformation of residual HCC after inadequate RFA treatment. In that study, activation of lncRNA-ASMTL-AS1 by Myc resulted in the downregulation of miRNA-342-3p, exacerbating HCC cell malignancy by activating nemo-like kinase (NLK)/YAP signaling. The authors proposed that lncRNA-ASMTL-AS1 may serve as a novel predictive and therapeutic target for residual HCC after RFA [88].
In prior research, expression of the LIM kinase (LIMK) family, which is characterized by modulation of the actin cytoskeleton, was associated with metastasis progression [113,114]. LIMK1 was identified as a miRNA-520-3p target. As a tumor suppressor, miRNA-520-3p inhibits HCC progression by downregulation of the LIMK1 axis. Notably, lncRNA H19 accelerates HCC proliferation and metastasis by increasing LIMK1 activation through miRNA-520-3P sequestration [89].
3. Exosomal circular RNAs as prognostic markers
Several exosomal circRNAs have been reported to be associated with target genes and miRNAs. Wang et al. [89] demonstrated that metastasis-related serum exosomal circPTGR1 is associated with advanced tumor clinical stage and poor prognosis via the miRNA-449a/MET pathway. Previous study have shown that overexpression of miRNA-449a, which functions as a tumor suppressor, inhibits tumor cell growth, migration, and invasion [115]. The proto-oncogene MET is one of the targets of miRNA-449a. circPTGR1 may play a crucial role in downregulating the interactions between miRNA-449a and MET, which can disrupt the homeostasis of the tumor microenvironment (TME) and enhance HCC progression. Considering that circPTGR1 is highly expressed in malignant metastatic HCC cells, it may serve as a prognostic and therapeutic marker in patients with HCC.
Zhang et al. [90] revealed that exosomal circular ubiquitin-like with plant homeodomain and ring finger domain 1 RNA (circUHRF1), generated in large amounts by HCC cells, induces natural killer (NK) cell dysfunction in HCC, thereby assisting with immunosuppression. Thus, it was demonstrated that circUHRF1 may contribute to resistance to anti-PD1 immunotherapy in patients with HCC. Gong et al. [91] showed that the circ-ZEB1.33-miRNA-200a-3p-cyclin dependent kinase 6 (CDK6) regulatory axis plays a role in the reinforcement of proliferation in human HCC; circ-ZEB1.33, which stimulates the growth of human HCC by targeting miRNA-200a-3p and elevating CDK6, was detected in tumors and serum, and its levels may be utilized to predict the outcomes of individual patients.
Huang et al. [92] demonstrated that exosomal circRNA-100338 promotes HCC metastasis by affecting cell proliferation, angiogenesis, and vessel permeability. Moreover, high expression of exosomal circRNA-100338 may serve as a prognostic factor for lung metastasis and poor survival in patients with HCC who have undergone curative hepatectomy. Exosomal circRNAs have been identified as tumor suppressors in HCC. Chen et al. [65] reported that exosomal circRNA-0051443 inhibited HCC progression by sponging miRNA-331-3p and regulating BCL2 antagonist/killer 1 (BAK1) expression. Previous studies have shown that BAK1 is a target of miRNA-331-3p and a regulator of mitochondria-mediated apoptosis, which is linked to the development of several cancers, including cervical and non-small cell lung cancers [116,117].
Therapeutic tools of exosomal noncoding RNAs in hepatocellular carcinoma
Recently, researchers have explored the use of recipient cell-derived exosomal ncRNAs as therapeutic treatments for HCC (Table 3 [88,90,118-126]). In a previous study, downregulation of exosomal miRNA-122 was linked to HCC development and progression [42]. Lou et al. [118] demonstrated that miR-122 delivered through adipose tissue-derived mesenchymal cell exosomes was transferred to recipient HCC cells, enhancing their sensitivity to systemic chemotherapy, including sorafenib. Similarly, Wang et al. [119] showed that exosomes derived from stellate cells loaded with miRNA-335-5p reduced HCC progression and metastasis and caused tumor shrinkage. Moh-Moh-Aung et al. [120] found a negative correlation between miRNA200b-3p and ETS-related gene (ERG), a gene associated with HCC angiogenesis. Additionally, exosomes from an HCC cell line overexpressing miRNA200b-3p were used to demonstrate the ability of miRNA200b-3p to decrease angiogenesis by reducing ERG expression. Using engineered exosomes designed to selectively bind to HepG2 cells, it was confirmed that upregulated miRNA-26a reduces the migration and proliferation capabilities of the cells [121].
Efforts are underway to investigate exosomal lncRNAs as potential therapeutic targets for HCC. Analysis of the miRNA-342-3p/NLK/YAP signaling pathway revealed a high level of exosomal ASMTL-AS1 expression in cases where HCC persisted after inadequate treatment with RFA. Consequently, regulating the expression of ASMTL-AS1 through lncRNA may serve as a potential therapeutic strategy for HCC [88].
Recent studies have indicated that circRNAs are associated with the immunosuppressive TME of HCC. Huang et al. [122] have revealed that circMET is upregulated in HCC tissues via the miRNA-30-5p/Snail/dipeptidyl peptidase 4 (DPP4)/CXCL10 pathway, which further exacerbates the immunosuppressive TME. Moreover, the combination of the DPP4 inhibitor sitagliptin and an anti-PD1 antibody has been shown to elevate the number of CD8+T lymphocytes infiltrating the tissue microarray [122]. Zhang et al. [90] demonstrated a positive correlation between the expression of circUHRF1 in the exosomes of HCC cells and poor clinical outcomes in patients with HCC. In addition, analysis of the molecular mechanisms of circUHRF1 in NK cells showed that a high level of plasma exosomal circUHRF1 was associated with a reduction in the NK cell proportion and tumor infiltration. Modulation of exosomal circUHRF1 demonstrated therapeutic potential in regulating the immunosuppressant TME in HCC cases resistant to anti-PD1 treatment [90].
Several natural compounds and their derivatives provide innovative possibilities for cancer treatment. Zhou et al. [123] found that HCC proliferation and metastasis are inhibited by gomisin M1 and its analogs, which target TAR-RNA binding protein to control miRNA synthesis, change the levels of a subset of miRNAs (such as miRNA-497-5p, miRNA-146a-5p, and miRNA-10b-5p), and affect pathways relevant to cancer. Tang et al. [124] reported that solamargine substantially reduced the production of mucin short variant S1 (MUC1) protein by upregulating the expression of miRNA-4726-5p, which is regulated by the lncRNAs HOTTIP and TUG1. Their study demonstrated that solamargine inhibited HCC growth and boosted the anticancer effect of sorafenib through the HOTTIP-TUG1/miRNA-4726-5p/MUC1 signaling axis. These findings suggest prospective therapeutic targets and approaches for HCC management. Li et al. [125] found that notoginsenoside R1 inhibits the PI3K/Akt pathway and exhibits anti-hepatoma activity by downregulating miRNA-21. Li et al. [126] demonstrated that oroxin B has efficient anticancer properties by upregulating the production of miRNA-221, which in turn causes the PI3K/Akt signaling pathway to be inactivated and liver cancer cells to undergo apoptosis. These findings reveal the possibility of antitumor effects in HCC. Zhang et al. [127] demonstrated that sanguinarine could stimulate the production of miRNA-16 in HCC cells with either wild-type or mutant p53, but not in cells with p53 eradicated. More notably, sanguinarine may cause cell cycle arrest and apoptosis, inhibiting the proliferation of tumor cells.
Further research is necessary to assess the role of tumor suppressors in the transport of exosomal lncRNAs and circRNAs. Investigating the correlation between target genes/miRNAs, exosomal lncRNAs, and circRNAs could lead to the development of novel therapeutic targets for the treatment of HCC.
Conclusion
This review provides details of exosomal ncRNAs, such as miRNAs, lncRNAs, and circRNAs, their target genes in patients with HCC, and their clinical applications. However, the clinical applications of serum exosomal ncRNAs in HCC are restricted by an inadequate understanding of ncRNA regulatory networks and the complexity of target gene associations. It is critical to establish the precise relationship between exosomal ncRNAs and their target gene networks using extensive and validated prospective data.
Owing to the abundance of exosomes in biofluids such as ascitic fluid, utilizing exosomal ncRNAs derived from ascitic fluid for the diagnosis and prognostic evaluation of HCC is promising, surpassing the limitations of conventional blood-derived exosomal ncRNAs. Given that radiological modalities have relatively low sensitivity for the early diagnosis of HCC, it would be worthwhile to explore the correlation between radiological imaging markers, exosomal ncRNAs, and the classic biomarker AFP. Exosomal ncRNAs have potential as biomarkers for early diagnosis and prognosis and as therapeutic tools in HCC.
Notes
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
Acknowledgements
The authors are grateful to Hyo Eun Lee, the design director (Institute of Medical Science, Yeungnam University).
Funding
This research was supported in part by grants from the Bio & Medical Technology Development Program of the National Research Foundation (NRF) of the Korean government (grant numbers: 2019M3E5D1A02068089 and 2021R1G1A1094767).
Author contributions
Conceptualization, Funding acquisition, Investigation: MKK; Data curation, Visualization: JSY, MKK; Writing-original draft: JSY, MKK; Writing-review & editing: JSY, MKK.



