Pathophysiology and protective approaches of gut injury in critical illness
Article information
Abstract
The gut is a complex organ that has played an important role in digestion, absorption, endocrine functions, and immunity. The gut mucosal barriers consist of the immunologic barrier and nonimmunologic barrier. During critical illnesses, the gut is susceptible to injury due to the induction of intestinal hyperpermeability. Gut hyperpermeability and barrier dysfunction may lead to systemic inflammatory response syndrome. Additionally, gut microbiota are altered during critical illnesses. The etiology of such microbiome alterations in critical illnesses is multifactorial. The interaction or systemic host defense modulation between distant organs and the gut microbiome is increasingly studied in disease research. No treatment modality exists to significantly enhance the gut epithelial integrity, permeability, or mucus layer in critically ill patients. However, multiple helpful approaches including clinical and preclinical strategies exist. Enteral nutrition is associated with an increased mucosal barrier in animal and human studies. The trophic effects of enteral nutrition might help to maintain the intestinal physiology, prevent atrophy of gut villi, reduce intestinal permeability, and protect against ischemia-reperfusion injury. The microbiome approach such as the use of probiotics, fecal microbial transplantation, and selective decontamination of the digestive tract has been suggested. However, its evidence does not have a high quality. To promote rapid hypertrophy of the small bowel, various factors have been reported, including the epidermal growth factor, membrane permeant inhibitor of myosin light chain kinase, mucus surrogate, pharmacologic vagus nerve agonist, immune-enhancing diet, and glucagon-like peptide-2 as preclinical strategies. However, the evidence remains unclear.
Introduction
The gut is a complex organ that carries out important functions including digestion, absorption, endocrine regulation, and immunity. Microscopically, the gut wall consists of the serosa, muscularis propria, submucosa, and mucosa. The mucosa consists of the epithelium, lamina propria, crypt of Lieberkühn, and so on. The gut is covered by an epithelial layer with a surface area of 30 m2. The size of the surface area is similar in size to half a badminton court [1]. The epithelium plays a critical role as the first line of protection against pathogens and is important for the management of host homeostasis. Additionally, it is the central coordinator of mucosal immunity [2]. The lamina propria serves as a protective layer against microorganisms and is rich in immune cells. The gut is a continuously renewing organ with the majority of cells turning over within 1 week [3]. Several intestinal cells including absorptive enterocytes, mucus-producing goblet cells, hormone-producing enteroendocrine cells, and tuft cells are differentiated from intestinal stem cells residing near the base of crypt of Lieberkühn [4,5].
As a result of constant microorganism exposure, the gut has significant immune function. Initially, several lymphoid cells in the lamina propria and Peyer’s patch can detect pathogenic antigens. From these lymphoid cells, afferent lymphatics are drained from the mesenteric lymph nodes. Secretory immunoglobulins (Ig) including secretory IgA and immune cells can subsequently inhibit the pathogenicity of the antigen. Notably, secretory IgA inhibits the adherence of bacteria to the epithelium and prevents their colonization and multiplication. In addition, secretory IgA neutralizes bacterial toxins and viral activity and blocks the absorption of antigens from the gut [6]. These complex immune responses, cellular reactions, and immune cascades in the gut are called mucosal barriers [6].
These mucosal barriers consist of immunologic and nonimmunologic barriers. The immunologic barrier consists of secretory IgA and IgM lymphocytes. The nonimmunologic barrier consists of digestive enzymes, mucus, peristalsis, and gut flora [6]. In animal studies, sepsis can induce a decrease in the crypt proliferation and a diminution of the villus length [7,8].
Mucus plays an important role in the mucosal defense by preventing the traversal of bacteria, digestive enzymes, and water-soluble toxic molecules into the mucosal surface [9]. In critical illnesses, the role of the mucus is compromised. Consequently, the damage to the mucus layer results in epithelial cell dysfunction. Gut ischemia/reperfusion leads to the loss of hydrophobicity of the mucus layer and altered intestinal permeability [9].
Under normal circumstances, approximately 40 trillion microorganisms reside within the gut. Gut microbiota can degrade dietary plant polysaccharides and proteins, to a small degree, by fermentation. The main end products of the fermentation are short-chain fatty acids (SCFAs) such as butyrate, acetate, and propionate. The SCFAs play an essential role in the maintenance of colonic integrity and metabolism. It has been shown that butyrate serves as the main energy source for colonocytes [10,11]. Several mechanisms regarding the maintenance of gut stability and homeostasis by microbiota have been proposed [12].
Preclinical studies have shown that the microbiome is essential for the protection against enteric and systemic pathogens through diverse mechanisms [13]. First, commensal microbiota can directly outcompete with intestinal pathogen or kill potential invaders by producing defensins and signaling molecules [14]. Second, microbiota are potent inducers of the immune system [15].
Pathophysiology of gut injury
During critical illnesses, it is commonly observed that the gut is susceptible to injury [16]. In the intensive care unit (ICU), approximately 50% of the patients experience enterocyte damage [17]. The gut has been hypothesized to be the “motor” of critical illness [18,19]. This theory is based on the fact that critical illness induces intestinal hyperpermeability, leading to bacterial translocation and subsequent systemic infection. Gut damage causes gastrointestinal (GI) symptoms. The GI symptoms caused by gut damage occur in approximately 62% of the patients in ICUs [17].
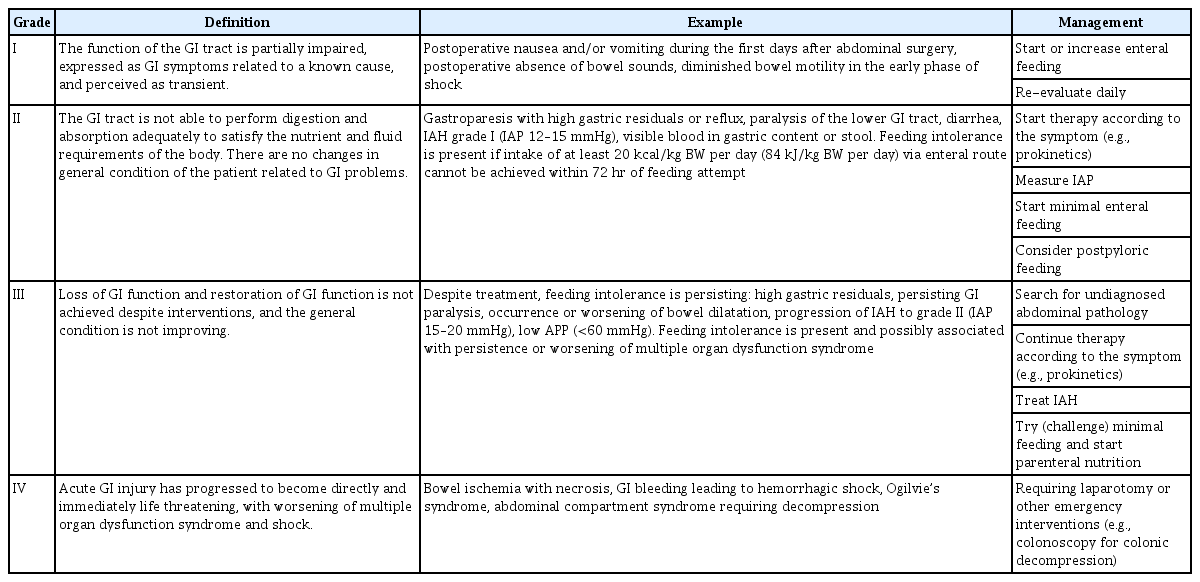
In the 1970s, a gut mucosal damage grading system in the shock state was proposed [20]. Recently, a GI dysfunction grading system for critically ill patients was developed by the Working Group on Abdominal Problem (WGAP) of the European Society of Intensive Care Medicine (ESICM) [21]. Acute GI injury (AGI) grade I refers to the development of new GI symptoms, such as vomiting, gastric residual volume, diarrhea, GI bleeding, paralysis of the lower GI tract, or abnormal bowel sounds related to a known cause and perceived as transient (risk of developing GI dysfunction or failure). AGI grade II refers to a lack of improvement in these symptoms and no change in the general condition. This grade is an indication for intervention (for example, prokinetics, postpyloric feeding) to restore the GI function. AGI grade III refers to the persistence of GI symptoms or worsening of multiple organ dysfunction syndrome and lack of improvement in enteral feeding. This means that interventions cannot restore the GI function. Lastly, AGI grade IV refers to the presence of acute life-threatening GI problems [21] (Table 1). Studies regarding these grading systems have shown that critically ill patients with GI dysfunction have higher mortality rates than patients without AGI [22].
During critical illness, excessive or inadequate adaptive responses evoked by intensive stress can have negative effects on the gut motility, mucosal blood flow, and mucosal permeability [19,23-26]. Gut hyperpermeability and barrier dysfunction may lead to a systemic inflammatory response syndrome, a clinical state that is also called “gut-derived sepsis” [27]. Sepsis induces a decrease in the crypt proliferation [7]. Critical illness induces gut mucosal hyperpermeability as early as 1 hour after the onset of sepsis and lasts at least 48 hours [3].
In addition, catecholamines administered to treat shock may lead to decreased microvascular perfusion in the gut [28]. This induces an increase of apoptosis and decrease of proliferation of small bowel mucosal cells, leading to the thinning of gut mucosa [29]. The resulting gut damage is exacerbated in the presence of chronic comorbidities such as cancer and chronic alcohol abuse [30,31]. Such injuries result in the impairment of the gut barrier and dysregulation of intestinal microbiota [32,33]. Therefore, small-intestinal mucosal integrity may be damaged in critically ill patients, leading to an increased intestinal permeability and intolerance to enteral nutrition [34].
Additionally, the gut microbiota is altered during conditions of critical illness. Within hours of the onset of a critical illness, the normal microbiota can convert to a disease-promoting pathobiome [35]. The diversity of a microbiome is significantly impaired during critical illness [3]. The etiology of such microbiome alteration in critical illness is multifactorial. These factors include an isolated host milieu, ancestral or newly expressed genes, numerous medical drugs, nutrients, and nutrition support routes [3]. In injured gut microbiota, diet is the key to shaping the ecosystem of the gut microbiome, which is important for host metabolism [36]. In critically ill patients, the microbiota can be severely altered to become unstable [37].
Critical illness leads to multiple changes to the microbiome, including the loss of diversity and overgrowth of pathogenic bacteria [38]. Distortion of the composition and diversity of the gut microbiome is defined as “dysbiosis.” Recently, it has been hypothesized that gut microbiome injury can cause distant organ injury. Microbiota-derived components such as pathogen-associated molecular patterns and metabolites derived from the gut can reach the circulatory system and interact at a systemic level to influence the immune homeostasis [39,40].
Such interactions or systemic host defense modulations between a distant organ and the gut microbiome is increasingly studied in disease research. Subsequently, the theory of gut-organ axes (gut-lung axis, gut-brain axis, gut-kidney axis, and gut-liver axis) has been developed [40].
1. Gut-lung axis
Evidence regarding the gut-lung axis emerged 20 years ago from rat model studies of trauma and hemorrhagic shock [41]. It was demonstrated that during trauma and hemorrhagic shock, gut-derived compounds translocated through the mesenteric lymph, causing distal lung injury [42].
2. Gut-brain axis
Recent research has shown that an extensive crosstalk between the gut microbiome and the brain through neuropeptides or endocrine processes, immune system signaling, and nerve signaling. The gut microbiome has been suggested to play a role in the cognitive function and behavior [43,44].
3. Gut-kidney axis
Recent murine studies have shown that therapy with SCFAs including acetate, propionate, and butyrate has protective effects in an ischemia/reperfusion model of acute kidney injury [45]. However, another study showed that the renal resident macrophages in the normal gut microbiome were sensitive to renal ischemia/reperfusion injury, resulting in initiation of inflammation and subsequent nephropathy after renal injury.
In contrast, the renal resident macrophages in a depleted gut microbiome are less sensitive to renal ischemia/reperfusion injury, resulting in an increased protection against renal ischemia/reperfusion injury [46].
4. Gut-liver axis
The liver may also be injured when the gut is injured. The liver is exposed to bacterial components and their metabolites via portal flow from the gut [47]. Therefore, the maintenance of gut mucosal integrity during critical illness is very important.
Protection of gut and several approaches to enhancing gut integrity and permeability
No treatment modality exists to significantly enhance the gut epithelial integrity, permeability, or mucus layer in critically ill patients. However, multiple helpful approaches including clinical and preclinical strategies exist. In animal studies, enteral nutrition can increase the blood flow in the gut during a “postprandial hyperemic response.” This may preserve the gut integrity and prevent gut-derived complications [48,49].
Enteral feed is associated with an increased mucosal mass and villus height in animal and human studies [50,51]. The trophic effects of enteral nutrition may help to maintain the intestinal physiology, prevent atrophy of gut villi, reduce intestinal permeability, protect against ischemia-reperfusion injury by stimulating intestinal perfusion, and preserve the gut immunity by affecting gut-associated lymphoid tissue [52]. However, the initiation of enteral nutrition should be started carefully considering enteral nutrition-derived complication, gut function, and contractility (e.g., ESICM WGAP recommendations) [53].
Several researchers have proposed that delayed trophic feeding (after 72 hours from intensive stress) is the optimal choice for critically ill patients with AGI, although this lacks evidence. As a protective strategy, trophic feeding may reduce the gut burden, help to maintain the intestinal physiology, prevent mucosal atrophy, and maintain the gut integrity in critically ill patients [54]. However, frequently providing enteral nutrition might have several complications, including vomiting, diarrhea, GI bleeding, aspiration pneumonia, refeeding syndrome, and gut ischemia [55,56]. In critically ill patients, enteral nutrition-related complications have been frequently observed [57].
According to surviving sepsis guidelines, enteral nutrition is recommended as soon as possible [58]. However, the tool for identifying patients who are likely to benefit from enteral nutrition among those who are critically ill with AGI and nutrition support protocol for decreasing enteral nutrition-related complications show poor performances [53].
The microbiome approach such as the use of probiotics, fecal microbial transplantation (FMT), and selective decontamination of the digestive tract has been suggested [3]. However, its evidence is not of high quality. Theoretically, the microbiome treatment will increase the number of “health-promoting” bacteria and decrease that of the “disease-promoting” bacteria. A microbiome treatment involves administering probiotics. Although significant evidence is not yet obtained, meta-analyses have demonstrated that ventilator-associated pneumonia improves following the administration of probiotics [59]. FMT involves the administration of an entire microbiome from a healthy donor and is considered for Clostridium difficile infections. Critically ill patients frequently receive antibiotics, and their microbiome is expected to alter due to antibiotic therapy. Therefore, FMT may be considered during critical illnesses [3]. However, the current evidence about microbiota-related therapies in critical illness remains unclear and limited to preclinical settings [40].
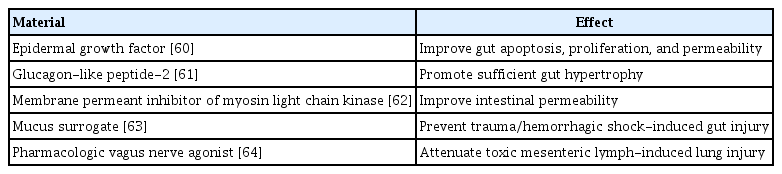
To promote rapid hypertrophy of the small bowel, several compounds have been reported, including epidermal growth factor, membrane permeant inhibitor of myosin light chain kinase, mucus surrogate, pharmacologic vagus nerve agonist, immune-enhancing diet, and glucagon-like peptide-2 as preclinical strategies [60-64]. However, the evidence remains unclear. Immune-enhancing nutrients (e.g., glutamine, alanine) can stimulate the enteric blood flow, maintain the mucosal barrier function by preserving tight-junction integrity, and induce the production and release of mucosal Ig and critical endogenous growth factors [6] (Table 2).
Conclusion
In critical illness, the gut is susceptible to injury due to multifactorial causes. No treatment modality exists to significantly enhance the gut barrier. Although current evidence is not of high quality, the enteral feed is associated with an increased gut barrier. Therefore microbiome treatment has been suggested. Additionally, in our review of the various studies, we concisely compiled the most up-to-date knowledge, on gut injury mechanisms and protection of the gut during critical illness. Further investigations should be performed with respect to the treatment of gut damage during critical illness.
Notes
Conflicts of interest
No potential conflicts of interest relevant to this article were reported.
Author contributions
Conceptualization: CYJ, JMB; Investigation and Resources: CYJ; Writing-original draft: CYJ, JMB; Writing-review & editing: JMB.
Previous presentations
The summary of this review was presented in the Acute and Critical Care Conference 2020 and the 40th Annual Meeting of Korean Society of Critical Care Medicine.